AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ir spectrum functional groups chart1/23/2024  
The exact position of this broad band depends on whether the carboxylic acid is saturated or unsaturated, dimerized, or has internal hydrogen bonding. The carbonyl stretch C=O of a carboxylic acid appears as an intense band from 1760-1690 cm -1. shows the spectrum of ethyl benzoate.įigure 10. The carbonyl stretch C=O of esters appears:įigure 10. shows the spectrum of butyraldehyde.įigure 9. If a compound is suspected to be an aldehyde, a peak always appears around 2720 cm -1 which often appears as a shoulder-type peak just to the right of the alkyl C–H stretches. 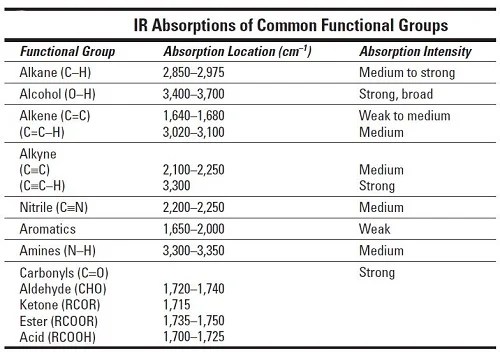
This is a saturated ketone, and the C=O band appears at 1715.įigure 8. C=O stretch - aliphatic ketones 1715 cm -1.The carbonyl stretching vibration band C=O of saturated aliphatic ketones appears: Note the very broad, strong band of the O–H stretch. O–H stretch, hydrogen bonded 3500-3200 cm -1įigure 7.Only alkenes and aromatics show a C–H stretch slightly higher than 3000 cm -1.įunctional Groups Containing the C-O BondĪlcohols have IR absorptions associated with both the O-H and the C-O stretching vibrations. This is a very useful tool for interpreting IR spectra. Note that this is at slightly higher frequency than is the –C–H stretch in alkanes. In aromatic compounds, each band in the spectrum can be assigned: 
The spectrum of 1-hexyne, a terminal alkyne, is shown below. In alkynes, each band in the spectrum can be assigned: As alkanes compounds, these bands are not specific and are generally not noted because they are present in almost all organic molecules. In alkenes compounds, each band in the spectrum can be assigned:įigure 4. Note that the change in dipole moment with respect to distance for the C-H stretching is greater than that for others shown, which is why the C-H stretch band is the more intense. Since most organic compounds have these features, these C-H vibrations are usually not noted when interpreting a routine IR spectrum. C–H rock, methyl, seen only in long chain alkanes, from 725-720 cm -1įigure 3.C–H bend or scissoring from 1470-1450 cm -1.In alkanes, which have very few bands, each band in the spectrum can be assigned: Hydrocarbons compounds contain only C-H and C-C bonds, but there is plenty of information to be obtained from the infrared spectra arising from C-H stretching and C-H bending. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
